Norepinephrine (also called noradrenaline) is a neurotransmitter produced by the locus coeruleus in the brainstem. It regulates alertness, attention, arousal, and the stress response, and works alongside dopamine in the prefrontal cortex to support executive function. Your prefrontal cortex needs norepinephrine in an optimal range — too little, and you can’t sustain focus, too much, and the brain shifts to reactive, survival-oriented processing.
In ADHD, norepinephrine signalling is dysregulated, contributing to difficulties with sustained attention, emotional regulation, and the stress response. Stimulant medications, NRIs and alpha-2 agonist medications all work partly by adjusting norepinephrine levels or activity in the brain.
Table of Contents[Hide][Show]
What is norepinephrine?
Norepinephrine (also known as noradrenaline) is a neurotransmitter produced by a tiny cluster of neurons in the brainstem called the locus coeruleus, and also by the adrenal glands that sit on top of your kidneys. It acts as both a neurotransmitter in the brain and a stress hormone in the body, and it is involved in an impressive range of things you do every day: staying alert, paying attention, responding to danger, regulating your heart rate, waking up, and calming down. 1 2
If you’ve read our dopamine entry, you’ll already know that dopamine and norepinephrine are closely related. Your body makes norepinephrine from dopamine — it’s the next step in the same production line. 3 They share a workplace (the prefrontal cortex, the bit behind your forehead that handles planning, decision-making, and impulse control), and they have similar hobbies. But where dopamine is primarily associated with motivation and reward, norepinephrine is more about alertness and vigilance. 4 They’re close relatives with overlapping social circles but different day jobs. 5
Norepinephrine (or, if it’s easier to remember, Nora Ephron — no, just kidding) is also the neurotransmitter that’s involved in half of what you experience as someone with ADHD. Waking up in the morning, waiting for your brain to boot up, but alertness takes its sweet time. When you are trying to fall asleep at night, hypervigilance is working overtime and just won’t switch off. A stressful e-mail can derail your entire afternoon. A long, intense conversation can lead to a crash and take hours to regulate from. That’s all norepinephrine doing its job, or at least trying to.
How dopamine becomes norepinephrine
Norepinephrine is built from dopamine. This manufacturing process explains a lot about why these two chemicals are so tightly connected — and why ADHD is never really a one-neurotransmitter problem.
Your brain builds norepinephrine through a chain of conversions, starting with an amino acid called tyrosine (found in protein-rich foods like eggs, cheese, meat, and soy). The chain looks like this: 6
Tyrosine → L-DOPA → Dopamine → Norepinephrine
Each arrow is an enzyme doing a specific job. The first step (tyrosine to L-DOPA, handled by an enzyme called tyrosine hydroxylase) is the bottleneck — it controls how much raw material enters the pipeline for both dopamine and norepinephrine. 6 If that first step slows down, everything downstream slows down with it.
The final step — the one that converts dopamine into norepinephrine — is handled by an enzyme called dopamine beta-hydroxylase (DBH). This enzyme needs specific cofactors to work properly, including vitamin C and copper. 3 This means norepinephrine production depends on dopamine being available and on having the right nutritional building blocks to convert it.
This is why we called them relatives earlier. Norepinephrine is dopamine’s literal biochemical descendant. They share a supply chain. When something disrupts dopamine production, norepinephrine production takes a hit too. And genetic variations in DBH — the enzyme at that final conversion step — have been linked to weaker executive function and poorer sustained attention in people with ADHD. 5
Science note: why “similar hobbies” is more than a metaphor
Dopamine and norepinephrine do overlap in what they do, but they do it in different places and in different ways. In the prefrontal cortex, norepinephrine strengthens the connections between neurons that are working on the same task (turning up the “signal”), while dopamine weakens the connections carrying irrelevant information (turning down the “noise”). 5 Together, they sharpen your brain’s ability to focus on what matters and ignore what doesn’t.
But their territory is different. Dopamine is concentrated in the striatum (involved in movement and reward) and the frontal cortex. Norepinephrine, via the locus coeruleus, projects to almost every region of the brain — cortex, amygdala, hippocampus, cerebellum, thalamus — with one notable exception: the basal ganglia, which is dopamine’s home turf. 1
So they share an office in the prefrontal cortex, but norepinephrine has a much wider beat. It’s less of a specialist and more of a roving correspondent, reporting to most of the brain at once.
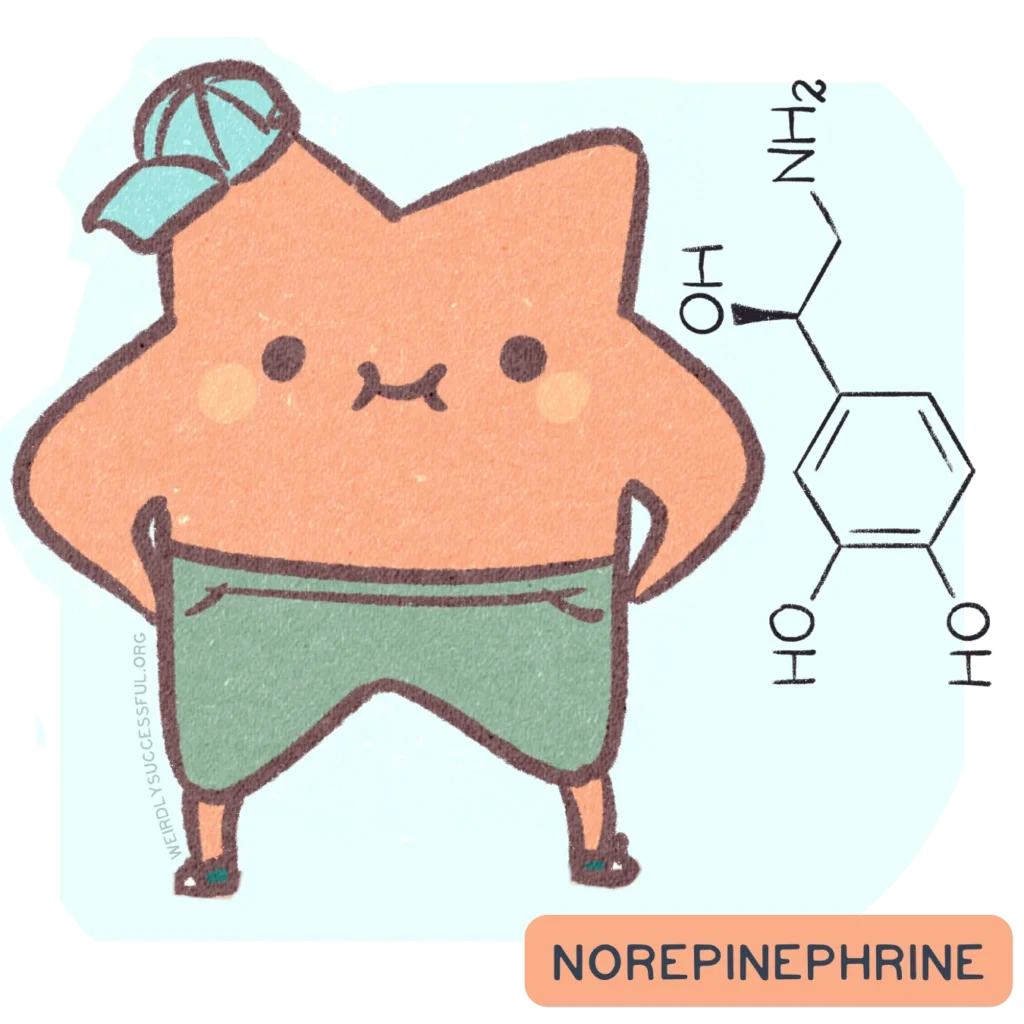
Group: Neurotransmitter and stress hormone (catecholamine family)
Also called: noradrenaline
Produced in: Locus coeruleus, plus adrenal glands
Built from: dopamine
Also known as: The alertness chemical. The one that decides whether you’re paying attention to the right thing or everything at once.
Role: Alertness, vigilance, arousal, attention regulation, stress response (fight-or-flight), blood pressure, heart rate, breathing rate, and mood regulation. Works alongside dopamine in the prefrontal cortex to support executive function.
Main system: The locus coeruleus projects widely across the brain — prefrontal cortex, amygdala, hippocampus, cerebellum.
Close relatives: Dopamine (its literal biochemical parent) and adrenaline/epinephrine (which is synthesised from norepinephrine). Norepinephrine is the middle generation in a three-generation family: dopamine → norepinephrine → adrenaline
Likes: Optimal levels in the prefrontal cortex (not too little, not too much — the same inverted-U curve as dopamine), adequate sleep, being allowed to do its job without chronic stress depleting the system
Dislikes: Chronic stress (burns through norepinephrine reserves), being confused with adrenaline, being overlooked in ADHD conversations, and the assumption that fight-or-flight is always a malfunction
ADHD medications targeting this system:
- Atomoxetine (Strattera) — a selective norepinephrine reuptake inhibitor.
- Guanfacine and Clonidine — alpha-2 adrenergic agonists that modulate norepinephrine’s effects in the prefrontal cortex. Stimulants (lisdexamfetamine, amphetamine) also increase norepinephrine alongside dopamine.
What norepinephrine does
Dials alertness
The locus coeruleus is tiny — roughly 50,000 neurons in humans, a speck of pigmented cells deep in the brainstem — but it projects to almost every region of the brain. 1 Think of it as a broadcast tower. When it fires, the signal reaches the prefrontal cortex, the amygdala, the hippocampus, the cerebellum, the thalamus. It’s how your brain sets its overall state of readiness.
The locus coeruleus has two modes of firing. The first is tonic — a steady, low-frequency background hum that sets your baseline level of alertness. The second is phasic — short, sharp bursts in response to something that matters. For example, a sudden noise, your name being called, or a notification that triggers a spike of “oh no, what now.” 1 7
These two modes create a kind of dial.
When tonic firing is low, you’re disengaged — zoned out, staring at a page without reading it, present in the room but not really in the room.
When tonic firing is moderate and phasic bursts are strong, you’re focused. The right things grab your attention and the wrong things don’t.
When tonic firing is high, you’re in scanning mode — restless, distractible, noticing everything and settling on nothing. 7
Why does this thing have two names?
Your adrenal glands sit on top of your kidneys. In Latin, “near the kidney” is ad renalis — which gives us adrenaline. In Greek, “upon the kidney” is epi nephros — which gives us epinephrine. Same gland, same chemical, in two different languages.
When scientists discovered the version of adrenaline that’s missing one methyl group, they added the prefix “nor-” (which is a derivation for “normal”, though there is an urban myth that it is short for the German N-ohne-Radikal, meaning “nitrogen without a radical”, but it is also applied to compounds that have no nitrogen at all). So nor-adrenaline in the Latin tradition, nor-epinephrine in the Greek one.
The result: the UK and most of Europe tend to say noradrenaline. The US tends to say norepinephrine. Research papers use both, sometimes in the same paragraph. They mean the same thing.
The one thing to remember: noradrenaline and adrenaline are not the same chemical. They’re related (one is made from the other), but they do different things. At least they don’t share the “nor-” part. That’s the one reliable way to tell them apart.
If that sounds like a description of your average weekday, you’re right. The locus coeruleus–norepinephrine system is one of the main reasons your ability to focus can swing between “completely absent” and “locked in” and “I can hear every conversation in this café, and I can’t concentrate on any of them” within the same afternoon.
Sharpens signals
One of norepinephrine’s most useful jobs is adjusting the contrast between what matters and what doesn’t. When the locus coeruleus fires a phasic burst, norepinephrine is released into sensory pathways across the brain. The effect is consistent: it suppresses spontaneous background activity while preserving or enhancing the response to whatever stimulus actually triggered the burst. 1
In practice, this means norepinephrine makes relevant signals crisper. In some neurons, it tightens the timing of spike responses, making the encoding of sensory information more precise. 1 In the auditory cortex, it reduces random firing while leaving the evoked response to a sound intact. 1 In the prefrontal cortex, it strengthens connections between neurons that are working on the same task. 5
The overall effect has been described as increasing “neural gain” — the brain’s equivalent of turning up the contrast on a screen. 8 When norepinephrine is doing this job well, you can pick out the voice you’re listening to in a noisy room, hold a train of thought while someone interrupts, or notice that your child has gone quiet (which, as any parent knows, is the most alarming sound of all). When it’s not doing this job well, everything arrives at the same volume.
Sara (2009) proposed that phasic norepinephrine also acts as a “network reset” — a signal that interrupts whatever pattern of brain activity is currently running and facilitates rapid reorganisation. 1 This is what allows you to shift your attention when something important changes. LC neurons respond most strongly to stimuli that are behaviourally relevant and unexpected, and their phasic responses precede changes in both forebrain activity and behavioural adaptation. 1 The locus coeruleus leads the transition.
The Goldilocks zone
If you’ve read our dopamine entry, the inverted-U curve will be familiar. Norepinephrine follows the same pattern: your prefrontal cortex needs an optimal amount, and either too little or too much impairs its function. 5
At the low end (when you’re drowsy, fatigued, or understimulated), there isn’t enough norepinephrine to support the prefrontal networks that handle planning, working memory, and impulse control. At the high end (when you’re stressed, panicked, or overstimulated), norepinephrine floods past the sweet spot and starts engaging a different set of receptors that actively weaken prefrontal function. 5
The receptor mechanics explain why the same chemical can help you focus and also make you fall apart.
At moderate levels, norepinephrine binds to alpha-2A receptors on prefrontal neurons. These receptors strengthen network connections — they turn up the signal, allowing the prefrontal cortex to maintain focus and guide behaviour. 5 This is the sweet spot. The Goldilocks zone.
At high levels (produced by stress, threat, or too much stimulant medication), norepinephrine starts engaging alpha-1 receptors instead. These do the opposite: they weaken prefrontal network connections, reduce working memory, and shift the brain toward more reactive, impulsive, survival-oriented processing. 5 9
Arnsten (2009) puts it like this: depleting norepinephrine and dopamine from the prefrontal cortex is as damaging to its function as removing the cortex entirely. 5 These chemicals are must-haves for our brains.
Science note: the receptor switch
Norepinephrine has a higher affinity for alpha-2A receptors than for alpha-1 receptors. This means that at moderate concentrations, it preferentially activates alpha-2A (which supports prefrontal function). Only when levels rise further does it begin engaging alpha-1 (which impairs it). 5 10
Under genuine threat, this is exactly what you want your brain to do — shift from slow, deliberate prefrontal processing to fast, reactive amygdala-driven processing. The problem in ADHD is that this switch can be triggered by threats that don’t warrant it — a critical email, an ambiguous text message, a tone of voice — and the prefrontal cortex goes offline at exactly the moment you need it most.
Fight, flight, and the rest of the alliterative family
You’ve probably experienced norepinephrine’s stress-response role firsthand. Someone is rude to you, or you sense danger, and you feel this urge to run away or to punch them in the face. Your heart rate spikes, your breathing changes, and your palms get sweaty. That’s norepinephrine (and its descendant, adrenaline) doing exactly what they were designed to do: preparing your body to respond to a threat. 11
(There are also other threat responses — friend, fawn, and freeze. Someone liked alliterations. But let’s not open that door today, because this entry is already going to be substantial, and I know you need to get back to whatever task you’re avoiding by procrastinating with reading useful stuff like this. Thanks, by the way. I’m honoured.)
At higher levels, norepinephrine activation produces the full fight-or-flight package: heightened arousal, increased attention to novel stimuli, impaired working memory, and the peripheral signs — increased heart rate, blood pressure and sweating. 11 12 When the threat passes, the system is supposed to settle. In ADHD, it often doesn’t settle cleanly, or it activates in response to things that aren’t actual threats.
This is where the “wired but tired” experience comes from. When norepinephrine and dopamine spike too high, the brain’s own wakefulness-promoting systems can be suppressed. The surface is buzzing — you feel agitated, restless, on edge — but the deeper arousal circuits have been overwhelmed. You’re overstimulated and exhausted at the same time. 2
Norepinephrine and sleep
The locus coeruleus is one of the brain’s most reliable wakefulness signals. Its neurons fire most during alert waking, less during light sleep, and fall almost completely silent during REM sleep. 2 13 At moderate levels, norepinephrine activity is simply incompatible with sleep. 2
This is relevant for ADHD in two directions. If the locus coeruleus doesn’t quiet down properly at night, sleep onset is delayed, which many ADHD adults will recognise as the “my brain won’t shut up” experience. And when the locus coeruleus has been firing hard all day (sustained effort, stressful situations, hyperfocus), it can develop a kind of functional fatigue, where norepinephrine release drops off sharply. 14 That’s the crash after a long, intense period of focus: your system ran hard, and now it needs to recharge, whether you’ve decided you’re done or not.
The locus coeruleus also sets the brain’s arousal threshold during deep sleep — how easily you can be pulled out of it, and how cleanly. Fluctuations in LC activity during NREM sleep determine whether you stay deeply asleep, surface into a brief microarousal, or transition into REM. 15 When that threshold is disrupted, the result can be partial awakenings where the body activates, but conscious awareness doesn’t fully come online. Night terrors — episodes of intense fear, rapid heart rate, and sometimes screaming or flailing during deep sleep — are one example of what a disordered arousal from NREM sleep can look like. 15 16
The push & pull of ADHD
Everything described above — the alertness dial, the signal sharpener, the Goldilocks zone, and the stress responses — converges on a single problem in ADHD: the same chemical that’s supposed to support your attention is also the one that drives your stress response. And in ADHD, it’s doing both jobs badly.
The research found that ADHD involves dysregulated norepinephrine signalling in prefrontal circuits. 4 5 But “dysregulated” means the system sits in the wrong place at the wrong time.
A large systematic review of 55 studies measuring autonomic nervous system function in ADHD found that when there was a group difference between ADHD and non-ADHD participants, hypo-arousal (too little activation) was the more common pattern. 17 Reduced skin conductance, smaller pupil responses, lower heart rate variability — the body’s readiness dial turned down. This was most consistent at rest and during tasks that required sustained attention — exactly the conditions where ADHD adults struggle most.
But the same review also found that the picture was mixed. Nearly half of all findings showed no group difference at all, and a smaller but real proportion showed hyper-arousal. 17 Other research has identified distinct biological arousal profiles within ADHD — some people consistently running underaroused, others consistently running overaroused, and others shifting between the two depending on context. 18
The same norepinephrine system that’s underperforming on Tuesday morning when you’re trying to focus on a spreadsheet is overperforming on Tuesday evening when someone makes a comment that sends you into fight-or-flight.
At rest or during boring tasks, norepinephrine levels in the prefrontal cortex sit too low — below the alpha-2A sweet spot, far from the Goldilocks zone. The prefrontal networks that support sustained attention, working memory, and impulse control don’t have enough signal to hold together. 5 You zone out. You lose the thread. You read the same paragraph four times.
Then something stressful happens, and norepinephrine surges past the sweet spot into alpha-1 territory. The prefrontal cortex goes offline, working memory collapses, and the brain shifts to reactive, survival-oriented processing — the fight-or-flight mode that makes you snap at a partner, fire off a regrettable email, or spend two hours spiralling about a comment that wasn’t even directed at you. 5 11
The prefrontal cortex goes offline at exactly the moment you need it most. And because the prefrontal cortex is also the part of the brain responsible for regulating your emotional responses, losing prefrontal function during stress impairs your thinking and your ability to manage the feeling that’s impairing your thinking.
Science note: the imbalance problem
Animal models of ADHD (spontaneously hypertensive rats) show something counterintuitive: the prefrontal cortex can be simultaneously hypodopaminergic (too little dopamine) and hypernoradrenergic (too much norepinephrine). 19 The problem is an imbalance — the ratio is wrong, and the wrong receptors are being activated. Treatments for ADHD aim to optimise the balance of catecholamine signalling in the prefrontal cortex, bringing the system back into the alpha-2A sweet spot. 5
Norepinephrine and anxiety
If the ADHD tension described above sounds like it should produce anxiety, that’s because it often does.
Anxiety disorders occur in roughly 25–50% of people with ADHD. 20 In adults, population studies report approximately five times higher odds of having an anxiety disorder if you have ADHD compared to if you don’t. 21 And having both conditions together makes everything harder — more severe symptoms, lower quality of life, higher rates of hospitalisation, and greater difficulty with daily functioning than either condition alone. 20 22
The relationship between the two isn’t a coincidence. Longitudinal research tracking children from preschool onwards found that early ADHD symptoms predicted later anxiety, but not the other way around. 23 Anxiety, in many cases, develops from living with a dysregulated norepinephrine system. It’s a downstream consequence — something that builds over time from living with a system that’s wired this way.
Norepinephrine in the prefrontal cortex at moderate levels supports working memory, impulse control, and the ability to evaluate whether a situation is actually threatening. At high levels, it shifts the brain into reactive, threat-oriented processing. 5 For someone whose norepinephrine system is already dysregulated — sitting too low during ordinary tasks and overshooting during stress — the threshold for tipping into that threat state is lower. A critical comment, an unexpected change of plan, or an ambiguous text message is enough for the system to flood past the sweet spot. The prefrontal cortex goes offline, and the amygdala takes over.
Over time, this pattern trains the brain to expect threat. Working memory deficits from ADHD make it harder to hold context (was that comment actually hostile, or was I misreading it?), which feeds uncertainty, which feeds anxiety, which further impairs working memory. 20 The cycle reinforces itself constantly.
This is also why ADHD and anxiety can be difficult to tell apart in a clinical assessment. Inattention that looks like ADHD can be driven by anxious rumination. Restlessness that looks like hyperactivity can be driven by chronic arousal. And the person sitting in the assessment room may have spent years attributing their difficulties to anxiety alone, because nobody ever mentioned norepinephrine. 24
For women and late-identified adults
The overlap between ADHD and anxiety is particularly relevant for women and people identified as neurodivergent later in life. Women with ADHD are more likely to present with inattentive symptoms and internalising difficulties — including anxiety and depression — rather than the externalising hyperactivity that historically drove referrals. 20 This means women are more likely to receive an anxiety diagnosis first and an ADHD diagnosis years or decades later, if at all.
Recent research has found that women show greater improvement than men on atomoxetine (a norepinephrine-targeting medication) across ADHD symptoms, including impulsivity and emotional dysregulation. 20 This positions atomoxetine as a particularly useful option when ADHD and anxiety co-occur — a point we’ll return to in the medication section.
Medications that target norepinephrine
Every effective ADHD medication increases norepinephrine signalling, dopamine signalling, or both in the prefrontal cortex. 5 The mechanism that all current ADHD medications share is that they move catecholamine levels in the prefrontal cortex closer to the alpha-2A sweet spot described earlier. They differ in how they do it, where their effects are strongest, and what side effects come along for the ride.
If you’ve spent time in ADHD communities, you’ll have heard the conversation focus almost entirely on dopamine. Stimulants are “dopamine drugs.” ADHD is a “dopamine deficit.” This is, however, only half the picture. Stimulants increase norepinephrine more than dopamine in the prefrontal cortex, and two entire classes of ADHD medication work primarily through the norepinephrine system with little or no direct effect on dopamine in reward pathways.
This section covers the three main classes. It is not medical advice and is not a substitute for a conversation with your prescriber. What it is meant to do is help you understand why your medication does what it does, so that the conversation can be more informed.
Stimulants: not just dopamine drugs
Methylphenidate (Ritalin, Concerta) and amphetamine-based medications (lisdexamfetamine – Vyvanse, Elvanse, and dexamfetamine – Amfexa, Celexa) are the most commonly prescribed ADHD treatments. They work by blocking the reuptake of both dopamine and norepinephrine, and in the case of amphetamines, also increasing their release. 4
For years, the research conversation focused on what stimulants do in the striatum — the deep brain structure involved in movement and reward. That’s dopamine territory. But more recent work using clinically relevant doses (rather than the much higher doses used in earlier animal studies) revealed something important: at therapeutic doses, stimulants preferentially increase catecholamine levels in the prefrontal cortex, with relatively little effect on subcortical reward areas. 5 And within the prefrontal cortex, they increase norepinephrine release more than dopamine release. 5
This revelation shifted the understanding of how stimulants actually work. Their therapeutic effect comes primarily from improving prefrontal cortex function — strengthening the top-down regulation of attention, behaviour, and impulse control. 5 The cognitive improvements are blocked by either norepinephrine alpha-2 receptor antagonists or dopamine D1 receptor antagonists, confirming that both systems are involved. 5
In the prefrontal cortex, there are very few dopamine transporters. Instead, the norepinephrine transporter (NET) clears both norepinephrine and dopamine in this region. 4 5 This is a quirk of the prefrontal cortex’s architecture that turns out to be clinically important: any drug that blocks the norepinephrine transporter will raise both norepinephrine and dopamine levels in the prefrontal cortex, even if it has no direct effect on the dopamine transporter.
One more detail from brain imaging: clinically relevant doses of methylphenidate significantly reduce norepinephrine transporter availability in the locus coeruleus, the raphé nuclei, the hypothalamus, and the thalamus. 4 The medication affects norepinephrine signalling across multiple brain regions that control arousal, stress, and sleep, which helps explain both its therapeutic effects and why timing and dose matter so much.
Science note: the dose curve
Stimulant medications follow the same inverted-U that norepinephrine itself follows. Therapeutic doses optimise prefrontal function by bringing catecholamine levels into the alpha-2A/D1 sweet spot. Higher doses push past the optimum, engaging alpha-1 receptors and impairing prefrontal function — producing the cognitive rigidity, emotional flatness, or “zombie” feeling that some people report at excessive doses. 5 The medication is working too well at increasing catecholamines past the point where the prefrontal cortex benefits. If stimulants turned you into a “zombie” or you feel blunted and numb, that doesn’t mean stimulants are not helping you. Talk to your prescriber, because lowering your dose and finding the sweet spot again could help.
Atomoxetine: the norepinephrine backup
Atomoxetine (Strattera) is a selective norepinephrine reuptake inhibitor (NRI). It blocks the norepinephrine transporter (NET) without directly affecting the dopamine transporter. 25 26
Atomoxetine, however, is usually somewhat less potent than stimulants, but is useful when stimulants are not suitable or when comorbidities are present that would rule out stimulants. It is a second-line treatment only if stimulants are not tolerated in any dose.
How atomoxetine works
Because of the prefrontal cortex quirk described above — where NET clears both norepinephrine and dopamine — atomoxetine raises both norepinephrine and dopamine levels approximately threefold in the prefrontal cortex. 25 5 But it does not increase dopamine in the striatum or nucleus accumbens (the reward areas). 25 This region-specific profile is why atomoxetine has low abuse potential compared with stimulants. The prefrontal cortex gets the catecholamine support it needs. The reward pathways are left alone.
In terms of what it does for the person taking it, atomoxetine improves response inhibition (the ability to stop yourself doing something once you’ve started), sustained attention, and working memory. 26 27 These effects are consistent with enhanced noradrenergic function in the prefrontal cortex.
Atomoxetine takes longer to reach full effect than stimulants (typically several weeks rather than hours), and its effect profile is different. Where stimulants provide a noticeable phasic boost — you feel them kick in and wear off — atomoxetine provides a steadier, tonic adjustment to baseline norepinephrine levels. This makes it a useful option when stimulants produce too many peaks and troughs, when anxiety is a concern (since it doesn’t spike dopamine in reward areas), or when substance use history makes stimulant prescribing complicated. 4 20
Recent research has found that women show greater improvement on atomoxetine than men across ADHD symptoms, including impulsivity and emotional dysregulation. 20
Alpha-2 agonists: working the receptor directly
Guanfacine and Clonidine take a different approach entirely. Instead of increasing norepinephrine levels in the synapse (as stimulants and atomoxetine do), they mimic norepinephrine by binding directly to alpha-2 adrenergic receptors in the prefrontal cortex. 5 28
This is like bypassing the supply problem and going straight to the lock. Instead of increasing the amount of key floating around (norepinephrine in the synapse), you install a key that’s already shaped to fit (a drug that activates the same receptor). 5
Guanfacine is the more targeted of the two. It is 15–60 times more selective for alpha-2A receptors (the ones that strengthen prefrontal network connections) than for alpha-2B and 2C subtypes. 28 29 This selectivity means it can improve attention and impulse control with relatively less sedation and blood pressure lowering than clonidine, which activates all three alpha-2 subtypes plus imidazoline receptors. 28 5
Guanfacine works directly at the receptor rather than depending on norepinephrine being available in the synapse. That means it can work even when catecholamine levels are very depleted, which makes it useful as an add-on when stimulants or atomoxetine alone aren’t doing enough. 5 Dr. Rege (2025) describes this as adding “tonic, not a spike” — augmenting the baseline rather than pushing for more peaks. 30
In practice, both medications reduce ADHD symptoms, including inattention, hyperactivity, and impulsivity, and both can be used alone or added to stimulants when the response is incomplete. 28 They’re particularly useful when tics, aggression, or emotional dysregulation are prominent features — and when stimulant side effects are a problem. 5 28
The main trade-off is sedation. Both medications can cause tiredness, especially when starting. Clonidine is more sedating and more likely to lower blood pressure. 28 Abrupt discontinuation of either (particularly clonidine) can cause rebound effects and should always be done with medical supervision. 28
TL;DR — what to remember about norepinephrine
This has been a lot of neuroscience. Let’s recap the main points.
Norepinephrine works in tandem with dopamine, but they have different jobs. Dopamine is primarily about motivation and reward. Norepinephrine is primarily about alertness, attention, and the stress response. Your body makes norepinephrine directly from dopamine; they share a workplace in the prefrontal cortex, and most ADHD medications affect both. But when ADHD conversations only mention dopamine, they’re leaving out half the story.
Too little is just as bad as too much. Your prefrontal cortex needs norepinephrine in a narrow optimal range — the Goldilocks zone. Too little and you can’t sustain attention, hold information in working memory, or get started on things. Too much and the prefrontal cortex goes offline, handing control to more reactive, survival-oriented brain systems. In ADHD, the system tends to sit too low during ordinary tasks and overshoot during stress, which is why you can feel unfocused and hypervigilant on the same day.
How to support your norepinephrine Goldilocks state
You can help yourself stay in (or return to) the Goldilocks zone. Some of these are things your prescriber can help with, and some are things you can do yourself:
- Medication timing and form matter. Long-acting formulations produce fewer peaks and troughs than immediate-release. Dosing early in the day works with your locus coeruleus’s natural rhythm rather than against it. If you’re crashing in the afternoon, the answer may not be a higher dose — it may be a smoother delivery. Talk to your prescriber.
- Sleep is norepinephrine maintenance. The locus coeruleus recharges during sleep. Chronic sleep deprivation depletes the system’s ability to produce and regulate norepinephrine effectively. 31 Protecting your sleep is one of the most direct things you can do for your norepinephrine system.
- The building blocks need to be there. Norepinephrine is built from dopamine, which is built from tyrosine, which comes from protein. The final conversion step also requires vitamin C and copper. 3 None of this replaces medication, but a diet consistently low in protein or key micronutrients makes the whole production line less efficient.
- Chronic stress is the system’s biggest drain — and for neurodivergent people, the baseline is already higher. Sustained norepinephrine output burns through the system’s reserves and keeps the prefrontal cortex pushed past its sweet spot. The scientific term for this cumulative wear and tear is allostatic load — the total toll on your body when stress-response systems are activated repeatedly without fully recovering. 32 For neurodivergent people, the load includes things that don’t look like “stress” from the outside: constant sensory filtering, masking, translating between your communication style and everyone else’s, and managing executive function demands in systems that weren’t designed for how your brain works. 33 You can’t eliminate all of that. But you can look for where the load can be reduced — fewer masking demands, better sensory environments, structures that work with your brain rather than against it — and those reductions protect your norepinephrine system along with everything else.
- Know your pattern. If you can recognise when you’ve tipped out of the Goldilocks zone in either direction (the flat, can’t-start-anything low end or the reactive, can’t-think-straight high end), you have a better chance of intervening before the spiral takes hold. This entry exists partly to give you the language for that recognition.






